Dysfunction of the immune system is central to disease progression in cancer and chronic viral infection. The Sen laboratory investigates the regulation of T cell dysfunction and explores epigenetic approaches for T cell engineering. Our work lies at the interface of human immunology, systems biology, and functional epigenomics – merging clinical observations with mechanistic mouse studies to develop novel therapeutic strategies.
We have found that the regulatory “circuitry” of dysfunctional T cells differs remarkably from functional T cells fighting off acute viruses. By comparing chronic viral infections and cancer, we showed that this altered epigenetic wiring is a fundamental adaptation to chronic diseases and cannot be rescued by current treatments. Therefore, improved mechanistic understanding of this altered regulation will be critically important for reversing cancer-associated immune dysfunction. We are also exploring radical new approaches where we can “tune” specific components of the circuitry in immune cells to fix their pathological state in cancer while preserving their physiological role in other contexts, so we that we can minimize unwanted side-effects in patients. The fundamental questions we try to answer through our work in the lab are:
How do T cells commit to exhaustion?

T cell exhaustion, like many cellular differentiation processes, develops with a continuum of phenotypic and functional intermediate states. This prompts the question, what are the earliest signaling pathways that drive commitment to an exhausted T cell phenotype? Our previous work showed that changes in chromatin accessibility during chronic infection clearly precede functional deficits and are far more extensive than changes in gene expression. We are now exploring how these early regulatory changes initiate an exhausted epigenetic state that eventually becomes fixed and mediates downstream functional deficits.
How does the specific tumor environment shape T cell exhaustion?
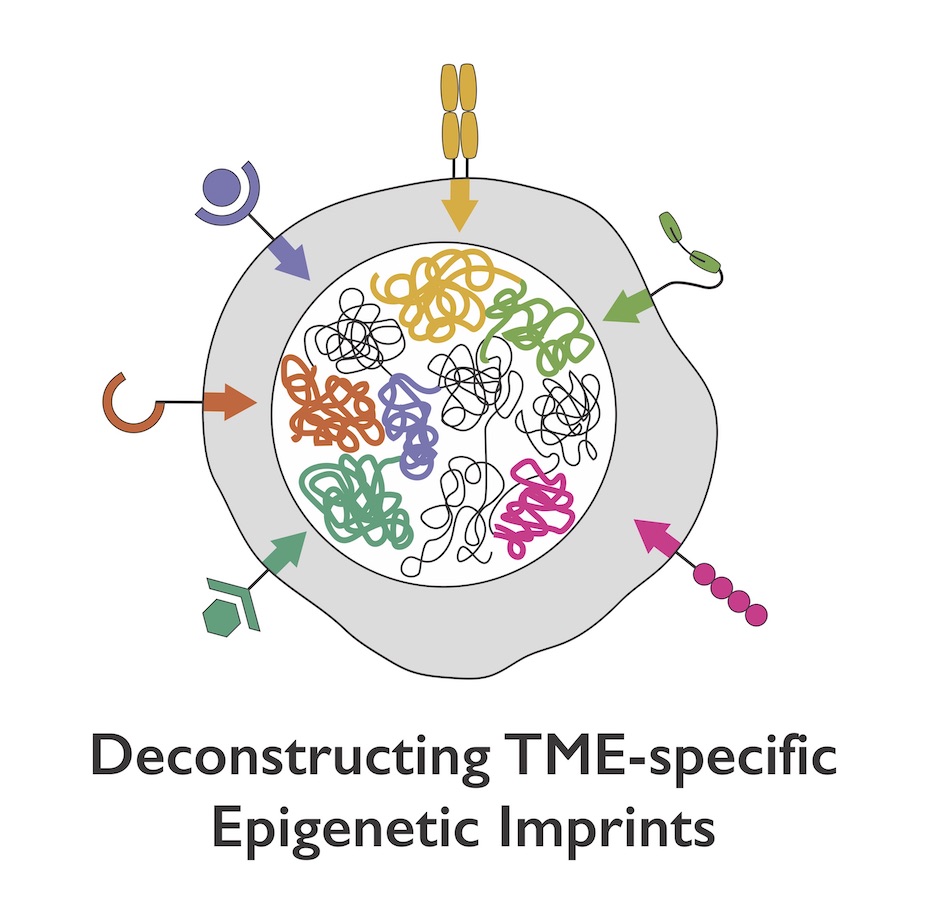
Despite a new recognition and interest in the biology of exhausted T cells as the targets of cancer immunotherapies, major questions remain about the impact of exhausted T cells in human tumors and the specific tumor contexts in which they arise. By mapping chromatin accessibility changes in several contexts of T cell exhaustion, our group has defined both common features of T cell exhaustion and disease-specific imprints of the unique microenvironment.
Critically, we have identified a core epigenetic signature, independent of disease-specific milieu, that can act as a precise biomarker of the exhausted state. We are now defining context-dependent differences in exhaustion that are induced by different mutations, treatment regimens etc. in immunotherapy-resistant tumors for rational prioritization of new therapeutic candidates.
How can we rescue exhausted T cells?

Our previous work suggests that many current therapeutic modalities, such as PD-1 checkpoint blockade in tumors or curative therapy in chronic infection, cannot reverse the epigenetic changes associated with exhaustion. Therefore, the most successful strategies to address T cell exhaustion must prevent the early establishment of this exhaustion program – for example, editing exhaustion-specific enhancers in T cells could be used to prevent chronic expression of inhibitory genes without knocking out the gene itself and so minimizing unwanted side effects. The Sen lab is empirically mapping the minimal regulatory features that can be targeted to avoid exhaustion and developing strategies for epigenetic reprogramming to complement immune checkpoint blockade therapy.
